Cervical Cancer Screening Guide
American Cancer Society (ACS) vs American College of Obstetricians and Gynecologists (ACOG)
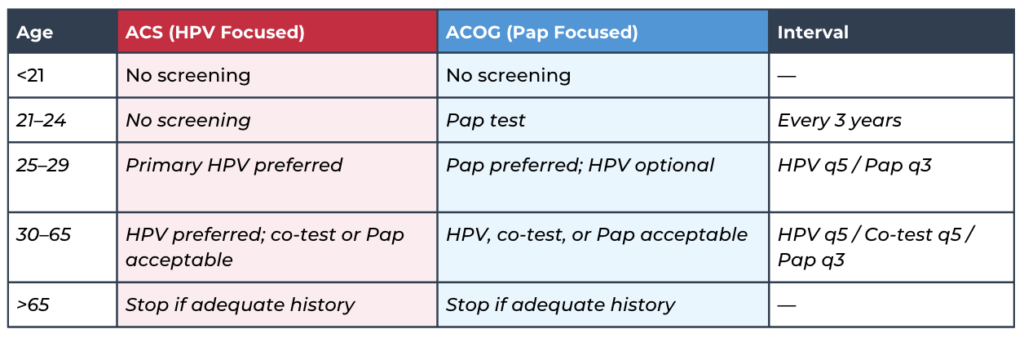
q3= 3 years and q5 = 5 years
Screening Methods (American Cancer Society - ACS):
- Primary HPV (preferred): Clinician-collected cervical sample, every 5 years.
- Self-collected HPV: FDA-approved kit, ordered by provider, every 3 years.
- Co-testing: HPV + Pap, every 5 years.
- Pap alone: Cytology only, every 3 years if HPV unavailable.
Laboratory Compliance Notes (CLIA/CAP):
- Use FDA-approved assays.
- Ensure documentation and provider communication.
- Align testing with ACS and ACOG guidelines.
Special Situations:
- Prior abnormal results or history of CIN2+ → follow risk-based management (ASCCP).
- Immunocompromised patients → require more frequent, individualized screening.
- HPV positive history → clinician-directed follow-up required.
- Self-collected HPV should NOT be used alone in high-risk patients.
View and download the informational PDF here.
References:
- American Cancer Society (ACS), 2020 Guideline Update: https://www.cancer.org/cancer/types/cervical-cancer/detection-diagnosis-
staging/cervical-cancer-screening-guidelines.html - American College of Obstetricians and Gynecologists (ACOG), 2021 Practice Advisory: https://www.acog.org/womens-health/faqs/cervical-cancer-screening